You have done some mixing and measuring & made graphs to calibrate the conductivity meter.... now we can use all that....
Does the tide affect the salinity of the water in the stream or the Maitai ?
Conductivity Calibration graph
Theory: notes etc (hmmm old site gone)
Theory: research task
Our 2016 data: HERE
Theory: notes etc (hmmm old site gone)
Theory: research task
Our 2016 data: HERE
Potato fish data
Salinity Investigation
Aim:
To carry out a practical investigation looking at the effect of the tide on the salinity of the stream or Maitai
Method
collect samples, measure conductivity (with the machine), calculate salinity (using the calibration graph) record your data
Note: we were collecting samples about 1 hour after high tide
Results
· Process your data:
- finish using the calibration graph to find the salinity of each sample (average where we have 2 readings)
- grab (Snipping Tool or similar) a Google earth image of the stream / Maitai and identify our sample locations
- add the salinity data to the Google image, show surface and bottom salinities at each site
· Research …. What is a salt wedge?
Figure out....How far down does the salt wedge go in our study area?
· Write your report, include:
Aim:
To carry out a practical investigation looking at the effect of the tide on the salinity of the stream or Maitai
Method
collect samples, measure conductivity (with the machine), calculate salinity (using the calibration graph) record your data
Note: we were collecting samples about 1 hour after high tide
Results
· Process your data:
- finish using the calibration graph to find the salinity of each sample (average where we have 2 readings)
- grab (Snipping Tool or similar) a Google earth image of the stream / Maitai and identify our sample locations
- add the salinity data to the Google image, show surface and bottom salinities at each site
· Research …. What is a salt wedge?
Figure out....How far down does the salt wedge go in our study area?
· Write your report, include:
- title
- aim
- method (maybe include some photos here too)
- results (our data processed and added to a Google map/image to show salinity)
- and discussion of the results (effect of tide, salt wedge?)
- add finished report to the dropbox

stream investigation
The problem of living in an area where the salinity changes
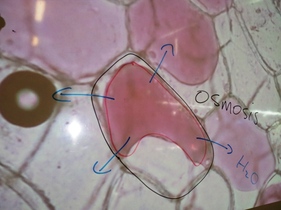
A red onion cell in salt water
Explain what has happened to it
Describe our potato fish experiment
Here is a link to our data and graph
Explain what has happened to it
Describe our potato fish experiment
Here is a link to our data and graph
Old website stuff:
What is a Mole
Just as:
one pair = 2 things
one dozen = 12 things
one score = 20 things
one gross = 144 things
one mole = 6.02 x 1023 things
that is...... 600 000 000 000 000 000 000 000
The number of people on earth is about 6 000 000 000
that is a heck of a lot less than a single mole !!!!!!
Q What chemical quantities contain one mole (ie 6.02 x 1023 ) things?
Ans The atomic mass (in grams) of a substance contains one mole of atoms
Moles ,n = grams of substance / atomic (or molecular) mass
n = g/Ar
Example:
H = 1 gram = 1 mole = 6.02 x 1023 atoms H
He = 4 grams = 1 mole = 6.02 x 1023 atoms He
Na = 23 grams = 1 mole = 6.02 x 1023 atoms Na
8 g of H is.....
8 g of He is.....
8 g of O is.....
Atomic and Molecular Weights
Atomic weight is the averaged mass of elements. They appear on the periodic table in the box for each element.
14
Si
28.08
Silicon
one mole of Si atoms = 28.08 g
Molecular weight or Molar Mass is the sum of the masses of all the atoms in a compound. It is the mass, in grams, of one mole of a compound.
Example: H2O, water. One mole contains:
1 mole of O atoms = 1 x 16 = 16 grams
2 moles of H atoms = 2 x 1 = 2 grams
So the molar mass of H2O = 16 + 2 = 18 grams
Example: Na2CO3, sodium carbonate. One mole contains:
1 mole of C atoms = 1 x 12 = 12 grams
2 moles of Na atoms = 2 x 23 = 46 grams
3 moles of O atoms = 3 x 16 = 48 grams
So the molar mass of Na2CO3 = 12 + 46 + 48 = 106 grams
Practice Calculating Molar Masses
Calculate the molar masses for each formula.
H2
CO2
NaCl
Na2SO4
C12H22O11
Cu(NO3)2
(NH4)2SO4
Or try this
http://www.chem.vt.edu/RVGS/ACT/notes/molar_mass.html
back to top Solutions
Concentration
This is the number of moles of the solute dissolved in one litre of the solution.
Concentration = no. of moles / volume (L)
Conc = n/L
Example 1: A 1 molar (1 M) solution of HCl contains 36.5 g of HCl per liter of solution, since 36.5 g is the molecular weight of HCl, 36.5 g/mole.
Example 2: A 0.5 molar (0.5 M) solution of HCl contains 18.25 g of HCl per liter of solution, since 18.25 g is half the molecular weight of HCl, 36.5 g/mole.
Q 1 Find the conc. if 5.8g of NaCl is dissolved in 1L of water?
Q 2 Find the conc. if 5.8g of NaCl is dissolved in 200mL of water?
Q 3 Find the conc. if 40g of NaOH is dissolved in 10L of water?
Q 4 Find the conc. if 0.40g of NaOH is dissolved in 100mL of water?
Q 5 How much NaCl is needed to give 500mL of a 0.25 mol/L solution?
Now let’s evaporate some sea water…..what for?
Is there a better way…I think so!
back to top
Salinity work.
Making up solutions for calibrating the conductivity meter
Now to our investigations....
Salinity of the coastal water 2007
Density
Seen one of these on a ship?
'Swimming' in the dead sea
back to top
Now to more investigations....
Salinity of the coastal water(Wed) 2007
Salinity of the coastal water(Thur) 2007
salt wedge... http://www.oceanservice.noaa.gov/education/kits/estuaries/media/supp_estuar05a_wedge.html
Maitai Channel salt wedge
(link removed for now)
back to top
What is a Mole
Just as:
one pair = 2 things
one dozen = 12 things
one score = 20 things
one gross = 144 things
one mole = 6.02 x 1023 things
that is...... 600 000 000 000 000 000 000 000
The number of people on earth is about 6 000 000 000
that is a heck of a lot less than a single mole !!!!!!
Q What chemical quantities contain one mole (ie 6.02 x 1023 ) things?
Ans The atomic mass (in grams) of a substance contains one mole of atoms
Moles ,n = grams of substance / atomic (or molecular) mass
n = g/Ar
Example:
H = 1 gram = 1 mole = 6.02 x 1023 atoms H
He = 4 grams = 1 mole = 6.02 x 1023 atoms He
Na = 23 grams = 1 mole = 6.02 x 1023 atoms Na
8 g of H is.....
8 g of He is.....
8 g of O is.....
Atomic and Molecular Weights
Atomic weight is the averaged mass of elements. They appear on the periodic table in the box for each element.
14
Si
28.08
Silicon
one mole of Si atoms = 28.08 g
Molecular weight or Molar Mass is the sum of the masses of all the atoms in a compound. It is the mass, in grams, of one mole of a compound.
Example: H2O, water. One mole contains:
1 mole of O atoms = 1 x 16 = 16 grams
2 moles of H atoms = 2 x 1 = 2 grams
So the molar mass of H2O = 16 + 2 = 18 grams
Example: Na2CO3, sodium carbonate. One mole contains:
1 mole of C atoms = 1 x 12 = 12 grams
2 moles of Na atoms = 2 x 23 = 46 grams
3 moles of O atoms = 3 x 16 = 48 grams
So the molar mass of Na2CO3 = 12 + 46 + 48 = 106 grams
Practice Calculating Molar Masses
Calculate the molar masses for each formula.
H2
CO2
NaCl
Na2SO4
C12H22O11
Cu(NO3)2
(NH4)2SO4
Or try this
http://www.chem.vt.edu/RVGS/ACT/notes/molar_mass.html
back to top Solutions
Concentration
This is the number of moles of the solute dissolved in one litre of the solution.
Concentration = no. of moles / volume (L)
Conc = n/L
Example 1: A 1 molar (1 M) solution of HCl contains 36.5 g of HCl per liter of solution, since 36.5 g is the molecular weight of HCl, 36.5 g/mole.
Example 2: A 0.5 molar (0.5 M) solution of HCl contains 18.25 g of HCl per liter of solution, since 18.25 g is half the molecular weight of HCl, 36.5 g/mole.
Q 1 Find the conc. if 5.8g of NaCl is dissolved in 1L of water?
Q 2 Find the conc. if 5.8g of NaCl is dissolved in 200mL of water?
Q 3 Find the conc. if 40g of NaOH is dissolved in 10L of water?
Q 4 Find the conc. if 0.40g of NaOH is dissolved in 100mL of water?
Q 5 How much NaCl is needed to give 500mL of a 0.25 mol/L solution?
Now let’s evaporate some sea water…..what for?
Is there a better way…I think so!
back to top
Salinity work.
Making up solutions for calibrating the conductivity meter
Now to our investigations....
Salinity of the coastal water 2007
Density
Seen one of these on a ship?
'Swimming' in the dead sea
back to top
Now to more investigations....
Salinity of the coastal water(Wed) 2007
Salinity of the coastal water(Thur) 2007
salt wedge... http://www.oceanservice.noaa.gov/education/kits/estuaries/media/supp_estuar05a_wedge.html
Maitai Channel salt wedge
(link removed for now)
back to top




